A certain weak acid has a dissociation constant of 1 × 10^-4 .The equilibrium for its reaction with a strong base is:

equilibrium - How to calculate the dissociation constant of a weak acid from the titration with a strong base? - Chemistry Stack Exchange
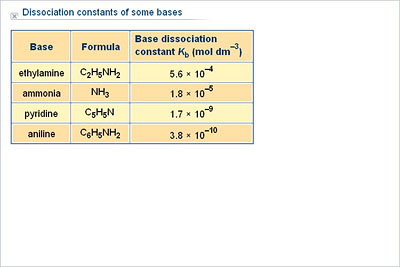
ACIDS AND BASES Dissociation Constants. weaker the acid, the stronger its conjugate base stronger the acid, the weaker its conjugate base. - ppt download
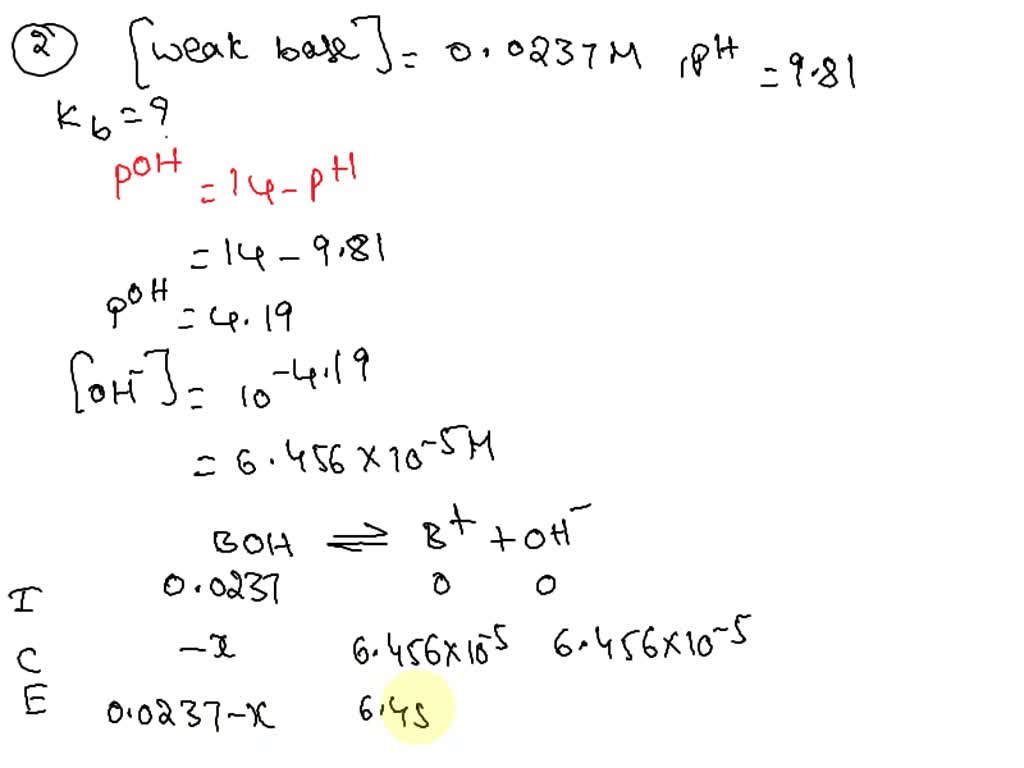
SOLVED: 1. The base-dissociation constant, Kb, for benzylamine is 3.88x10-10. What is the acid-dissociation constant, Ka for the benzylammonium ion? 2. A 0.0237 M solution of weak base is found to have

Question Video: Identifying the Correct Equation for the Base Dissociation Constant of a Generic Weak Base | Nagwa
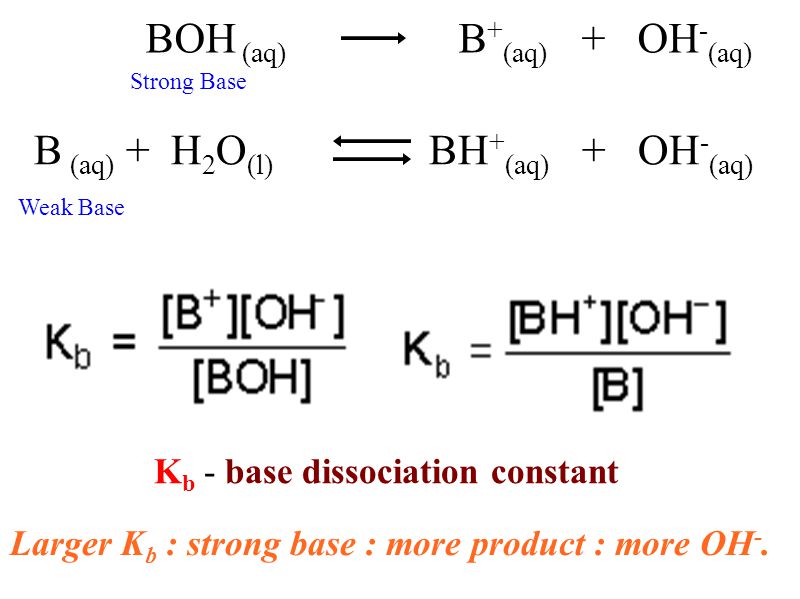
At 25°C, the dissociation constant of a base, BOH, is 1.0 x 10^-12. - Sarthaks eConnect | Largest Online Education Community


:max_bytes(150000):strip_icc()/what-is-pka-in-chemistry-605521_FINAL2-9fdfc39e9aa34caa96d6e74a2c687707.png)